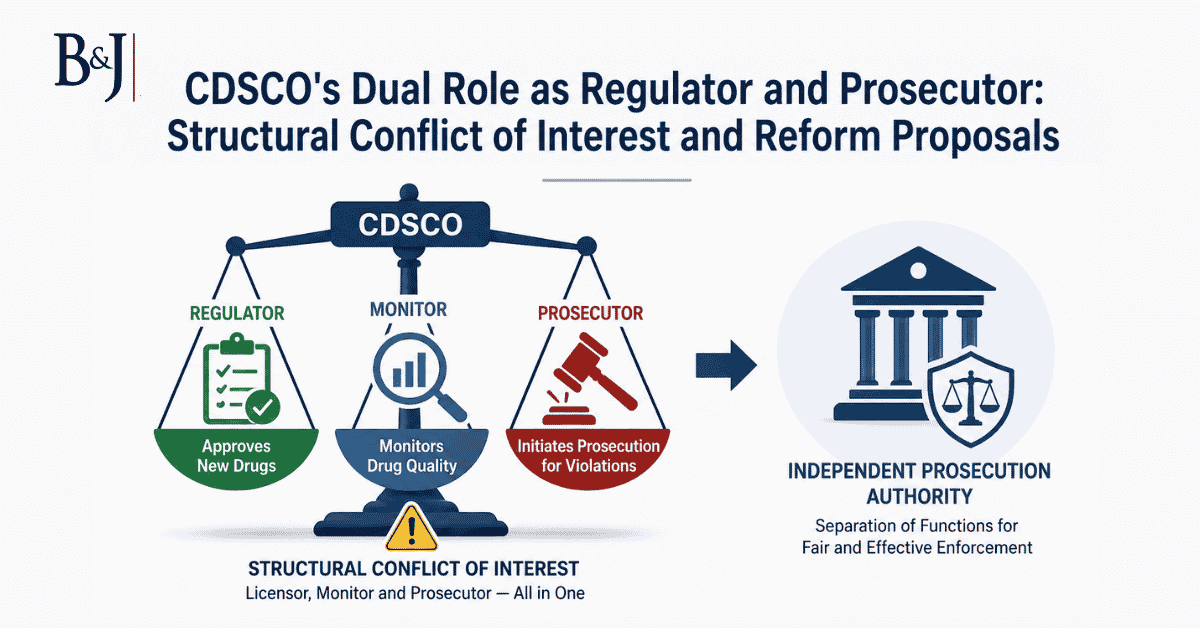
CDSCO’s Dual Role as Regulator and Prosecutor: Structural Conflict of Interest and Reform Proposals
ABSTRACT The Central Drugs Standard Control Organisation (CDSCO) occupies an unusual institutional position in India’s regulatory landscape: it is simultaneously the authorit
Strict Liability Offences Under the Drugs and Cosmetics Act, 1940: Are India’s Courts Adequately Enforcing Them?
ABSTRACT The Drugs and Cosmetics Act, 1940 creates strict liability offences for the manufacture and sale of not-of-standard-quality (NSQ) drugs. Parliament deliberately excluded t
Section 33P of the Drugs and Cosmetics Act: India’s Unused Instrument for Uniform Drug Law Enforcement
ABSTRACT Section 33P of the Drugs and Cosmetics Act, 1940 empowers the Central Government to give binding directions to State Governments for carrying into execution any provision
The Drug Inspector as the Sole Prosecution Authority: Why Police Cannot File FIRs Under Chapter IV of the Drugs and Cosmetics Act, 1940
ABSTRACT The Supreme Court’s landmark ruling in Union of India v. Ashok Kumar Sharma, (2021) 12 SCC 674 settled a long-contested question: police officers cannot register FIR
The Binding Nature of Administrative Instructions in India: A Unified Doctrinal Framework
ABSTRACT Indian governance is increasingly conducted through instructions, circulars, guidelines, and policy documents that are not enacted as formal legislation but profoundly sha
Compounding of Offences Under the Drugs and Cosmetics Act: The 2025 Rules and Their Interface with Prosecution
ABSTRACT The Drugs and Cosmetics (Compounding of Offences) Rules, 2025, notified on April 24, 2025 under Sections 32B and 33(2)(r) of the Drugs and Cosmetics Act, 1940, represent a
Can CDSCO Ignore Its Own Prosecution Guidelines? Legal Validity and Article 14 Challenge Explained
ABSTRACT The DCC and CDSCO prosecution guidelines are not legally binding as a matter of positive law — they cannot override the strict liability provisions of the Drugs and Cosm
Section 26A of the Drugs and Cosmetics Act, 1940: India’s Sharpest Drug Regulatory Instrument
ABSTRACT Section 26A of the Drugs and Cosmetics Act, 1940 empowers the Central Government to prohibit the manufacture, sale, or distribution of any drug in the public interest by n
The Mandatory Procedure for a Drug Inspector’s Complaint Under the Drugs and Cosmetics Act, 1940
ABSTRACT The Drugs and Cosmetics Act, 1940 establishes a unique prosecution architecture: criminal proceedings for quality offences can be initiated only through a complaint, not t
Section 119 IT Act vs Section 7 D&C Act: Why CBDT Circulars Are Binding in Nature But DCC Guidelines Are Not
ABSTRACT Two of India’s most prominent regulatory bodies — the Central Board of Direct Taxes (CBDT) and the Drugs Consultative Committee (DCC) — issue prosecution-related gui
 Whatsapp
Whatsapp